Lumefantrine established as the first line treatment for uncomplicated malaria, have been accompanied by the selection of polymorphisms associated with decreased lumefantrine sensitivity and by decreased ex vivo lumefantrine sensitivity. Reduced sensitivity to ACT partner drugs may exacerbate selection of artemisinin resistance, jeopardizing our most important antimalarial therapies. UNC669 recent work attributed the artemisinin resistance phenotype found in Southeast Asia to mutations in PF3D7_1343700, which encodes a protein homologous to kelch proteins from other organisms. Parasites selected for resistance to artemisinin demonstrated multiple mutations predicted to be in propeller domains of the protein. The mutations were associated with improved parasite survival after pulses of dihydroartemisinin, an in vitro correlate of artemisinin resistance, and with delayed clearance after artemisinin therapy in Cambodia. Specifically, the M476I mutation was selected in vitro in a Tanzanian parasite by longstanding cyclic artemisinin pressure, and 3 additional polymorphisms prevalent in Cambodian field isolates were associated with delayed clearance after therapy. The cysteine protease falcipain-2 is a principal P. falciparum hemoglobinase. Inhibition of this protease or knockout of the gene blocked hemoglobin hydrolysis in trophozoites and led to decreased artemisinin activity, as hemoglobin is required for a potent antimalarial effect. Interestingly, parasites selected in vitro for artemisinin resistance had a nonsense mutation at codon 69 of the FP2 gene, suggesting that parasites partially blocked hemoglobin processing to limit toxicity from artemisinin. Although ACT remains highly efficacious for the treatment of falciparum malaria and delayed parasite clearance after ACT has not been noted in Uganda, it was important to characterize the diversity of genes in which polymorphisms may contribute to artemisinin resistance. Our goals were to characterize the diversity of the K13 and FP2 genes and to determine if artemisinin selective pressure or relative delays in parasite clearance after therapy were associated with particular genotypes. We therefore sequenced these genes in P. falciparum isolates collected from Ugandan children under varied selective pressure from recent therapy with ACTs. Artemisinin resistance, manifested as delayed parasite clearance and correlated with diminished action of pulses of 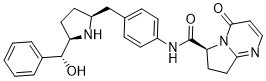 artemisinins in vitro, has recently been identified in Southeast Asia and associated with mutations in the regions of the P. falciparum K13 gene that encode the propeller domains. To characterize potential resistance markers in isolates from Uganda, we surveyed K13-propeller polymorphisms in recent isolates under varied levels of selective pressure due to prior therapy with ACTs. We identified limited diversity within the K13 gene, and did not detect any of the polymorphisms associated with artemisinin resistance in Southeast Asia. In addition, we found that the prevalences of K13-propeller polymorphisms identified in Uganda were not associated with recent use of ACTs or with the persistence of parasites $2 days following treatment with ACTs. Prior studies demonstrated that parasites treated with a FP2 inhibitor and FP2 deletion mutants were protected against an artemisinin pulse in vitro, indicating that the hemoglobinase FP2 is necessary for optimal activity of artemisinins. Interestingly, parasites selected in vitro for artemisinin resistance contained a stop mutation in the FP2 gene. To Tulathromycin B evaluate FP2 polymorphisms in Uganda over time.
artemisinins in vitro, has recently been identified in Southeast Asia and associated with mutations in the regions of the P. falciparum K13 gene that encode the propeller domains. To characterize potential resistance markers in isolates from Uganda, we surveyed K13-propeller polymorphisms in recent isolates under varied levels of selective pressure due to prior therapy with ACTs. We identified limited diversity within the K13 gene, and did not detect any of the polymorphisms associated with artemisinin resistance in Southeast Asia. In addition, we found that the prevalences of K13-propeller polymorphisms identified in Uganda were not associated with recent use of ACTs or with the persistence of parasites $2 days following treatment with ACTs. Prior studies demonstrated that parasites treated with a FP2 inhibitor and FP2 deletion mutants were protected against an artemisinin pulse in vitro, indicating that the hemoglobinase FP2 is necessary for optimal activity of artemisinins. Interestingly, parasites selected in vitro for artemisinin resistance contained a stop mutation in the FP2 gene. To Tulathromycin B evaluate FP2 polymorphisms in Uganda over time.
The FP2 gene in isolates collected before and after the widespread implementation of ACT
Leave a reply